|Intro| - |Sizes| - |Tour| - |Help|
[Quarks] - [Nucleons] - <Nucleus> - [Atom] - [Cell] - [People]
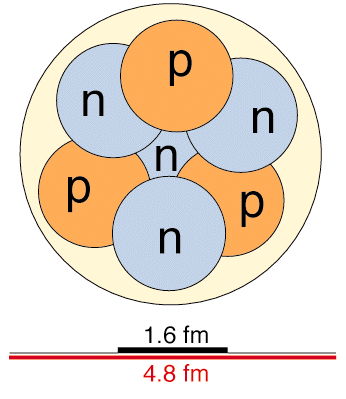
There is a very strong and very short-range force that pulls nucleons toward each other, and an even stronger repulsive force that keeps them from overlapping each other. The result is that a nucleus 'looks' like a closely packed set of spheres that are almost touching one another.

Natural Lithium is made up of two isotopes: Lithium-7 (92.5%) and Lithium-6 (7.5%). Lithium-6 has 3 protons (as it must if it is to be the element Lithium) but only 3 neutrons, so it has a mass number of 6.
The atoms of these two isotopes behave almost exactly the same way when doing chemistry, since the atomic electrons only care about the charge of the nucleus, but the physical properties of these two nuclei are very different. For example, the nucleus of Lithium-7 is not spherical, as the diagram above suggests, but deformed into a football-like shape as shown below.

The nucleus does not really have a sharp surface like this picture suggests. The protons and neutrons move around and there is a fuzzy quantum-mechanical probability of finding them outside the region shown with a shiny surface. We made this picture to show where th nucleons will be found most (more than 90%) of the time.
If we look more closely, we will notice that the protons and neutrons (nucleons) are made up of quarks interacting via gluon exchange. This is shown schematically in the picture below.
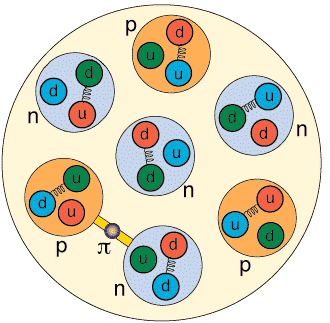
Nuclear physicists can do experiments that look inside the nucleus to see the protons and neutrons, and they can also do experiments that allow them to see the role of quarks and gluons in nuclei.